Phosphorus was made in 1669 by German scientist, Hennig Brand, and is a non-metallic. The word phosphorus is derived from the Greek term phosphoros. This means light bearing. It is used mostly in fertilizers because phosphorus is essential to all living things. Phosphorus is the second most important element to growing crops (nitrogen is first). Phosphorus appears in two common forms, white and red phosphorus.
|
WHITE
White phosphorus is a soft solid with a waxy appearance. This type of phosphorus is flammable and highly reactive and explosive in certain cases. White phosphorus also glows in the dark and is poisonous. If skin comes in contact with white phosphorus, severe burns can occur. White Phosphorus must be stored under water because it can ignite when in contact with air.
|
RED
Red phosphorus is a in a powdery form (still solid). Red phosphorus is much more stable then white phosphorus. This type of phosphorus is not poisonous like white phosphorus is. To change white phosphorus into red phosphorus, the white phosphorus must be exposed to sunlight.
|
FACTS & DEFINITIONS
|
Atomic Number= 15
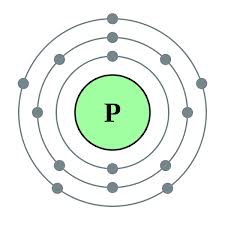
Number of protons in nucleus Atomic Mass= 30.97 Total mass of an atom Atomic Symbol= P Representation of the element Protons=15 Particles with positive charge Electrons=15 Particles with negative charge Neutrons=16 Particles with no charge Density= 1.8 grams/cm^3 Mass per unit Atomic Radius= 110 pm Measure from nucleus to farthest electron cloud Ionization Energy 1= 1061.339 kJ/mol Ionization Energy 2= 1895.937 kJ/mol Ionization Energy 3= 2909.612 kJ/mol Minimum amount of energy to get rid of an electron in the specific orbital |
Isotopes= 1
Different number of neutron possibilities Electronegativity= 2.1 Tendancy to attract electrons rated 0-4 Melting Point= 44.15° C When the element turns into a liquid state Boiling Point= 280.5° C When the element turns into a gaseous state Room Temperature Form= solid The form the element is at average room temperature Group= 15 (Pnictogen) Columns of elements on Periodic Table Period=3 Rows of elements on Periodic Table Classification= Non-metal Non-metals are elements with partially filled orbitals |
Electron Configuration= [Ne] 3s2 3p3
Bohr Diagram
Lewis Dot Diagram
Let's see if you were paying attention. Click the button below to take the test!