QUIZ TIME!
Were you paying attention?
1) FIll in the blanks...Phosphorus is a non-metal in group __ and period__.
2) Multiple choice... How many protons electron and neutrons are in Phosphorus?
a) 15 15 15
b) 16 15 15
c) 15 16 15
d) 2000 5000 78976
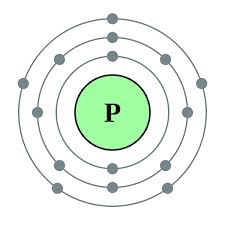
3) Short Answers... Describe the similarities and differences between the diagrams below. What are the names of the diagrams?
1) FIll in the blanks...Phosphorus is a non-metal in group __ and period__.
2) Multiple choice... How many protons electron and neutrons are in Phosphorus?
a) 15 15 15
b) 16 15 15
c) 15 16 15
d) 2000 5000 78976
3) Short Answers... Describe the similarities and differences between the diagrams below. What are the names of the diagrams?
Highlight below to see the answers
1) 15; 3
2) c
3) The Lewis Dot Diagram on the right shows the number of protons/neutrons in the farthest electron cloud. In the Bohr Diagram to the left shows all the protons/neutrons in the 1st 2nd and 3rd electron clouds. The Lewis Dot Diagram has 5 dots showing 5 protons/neutrons. If you look on the Bohr Diagram, you will see 5 protons/electrons in the farthest electron cloud. The difference between the two graphs is the Bohr Diagram shows ALL the protons/neutrons in Phosphorus (15).
1) 15; 3
2) c
3) The Lewis Dot Diagram on the right shows the number of protons/neutrons in the farthest electron cloud. In the Bohr Diagram to the left shows all the protons/neutrons in the 1st 2nd and 3rd electron clouds. The Lewis Dot Diagram has 5 dots showing 5 protons/neutrons. If you look on the Bohr Diagram, you will see 5 protons/electrons in the farthest electron cloud. The difference between the two graphs is the Bohr Diagram shows ALL the protons/neutrons in Phosphorus (15).